
San Diego, USA – April 17–22, 2026 – Suzhou Evopoint Biosciences Co., Ltd. (hereinafter referred to as "Evopoint") presented the latest preclinical data of its three core pipeline assets- XNW34017 (AURKA-MYC protein degrader), XNW27011 (CLDN18.2 ADC), and XNW28012 (TF ADC)- via poster presentation at the annual meeting of the American Association for Cancer Research (AACR 2026), one of the world’s largest cancer research conferences, showcasing the company’s cutting-edge innovation in oncology.
The AACR Annual Meeting serves as the definitive benchmark for global cancer research. In AACR 2026, multiple studies from Evopoint were selected for presentation, underscoring not only the international recognition of the company’s innovative prowess, but also the strategic foresight and robust R&D capabilities in drug discovery.
XNW34017 (AURKA-MYC degrader)
Poster Title: Discovery and Characterization of XNW34017, a First-In-Class, Orally Bioavailable, and Brain-Penetrant AURKA–MYC Degrader,#5157
Highlights: Aurora kinase A (AURKA), a serine/threonine kinase key to the mitotic progression, is frequently overexpressed in a broad spectrum of solid tumors and hematologic malignancies. While targeting its kinase activity has shown robust preclinical efficacy, the clinical translation of AURKA inhibitors has remained largely elusive up to date. In addition to its catalytic function, AURKA also serves as a critical molecular scaffold stabilizing MYC family protein, shielding them from proteasomal degradation. Unlike inhibitors that merely suppress AURKA kinase activity, AURKA degraders facilitate protein depletion of AURKA, effectively eliminating both its catalytic and scaffolding functions, thus releasing Myc family proteins to be degraded via proteasomal pathway. This dual-action mechanism of AURKA offers the potential for superior anti-tumor potency and an optimized therapeutic window, establishing AURKA-MYC protein degradation as a highly compelling therapeutic strategy.
Built upon this rationale, Evopoint has developed the world's first bifunctional AURKA-MYC degrader which can facilitate the highly efficient degradation of AURKA and Myc family proteins (c-Myc, L-Myc, and N-Myc) in Myc driven cancer cell lines. MOA studies of early lead compounds revealed that the depletion of AURKA strictly dependents on the target, the E3 ligase, and the ubiquitin-proteasome system (UPS). Furthermore, qPCR assays confirmed that the observed protein decrease of AURKA and Myc proteins were not caused at transcription level. These findings further validated the therapeutic rationale of inducing MYC degradation through the targeted depletion of AURKA.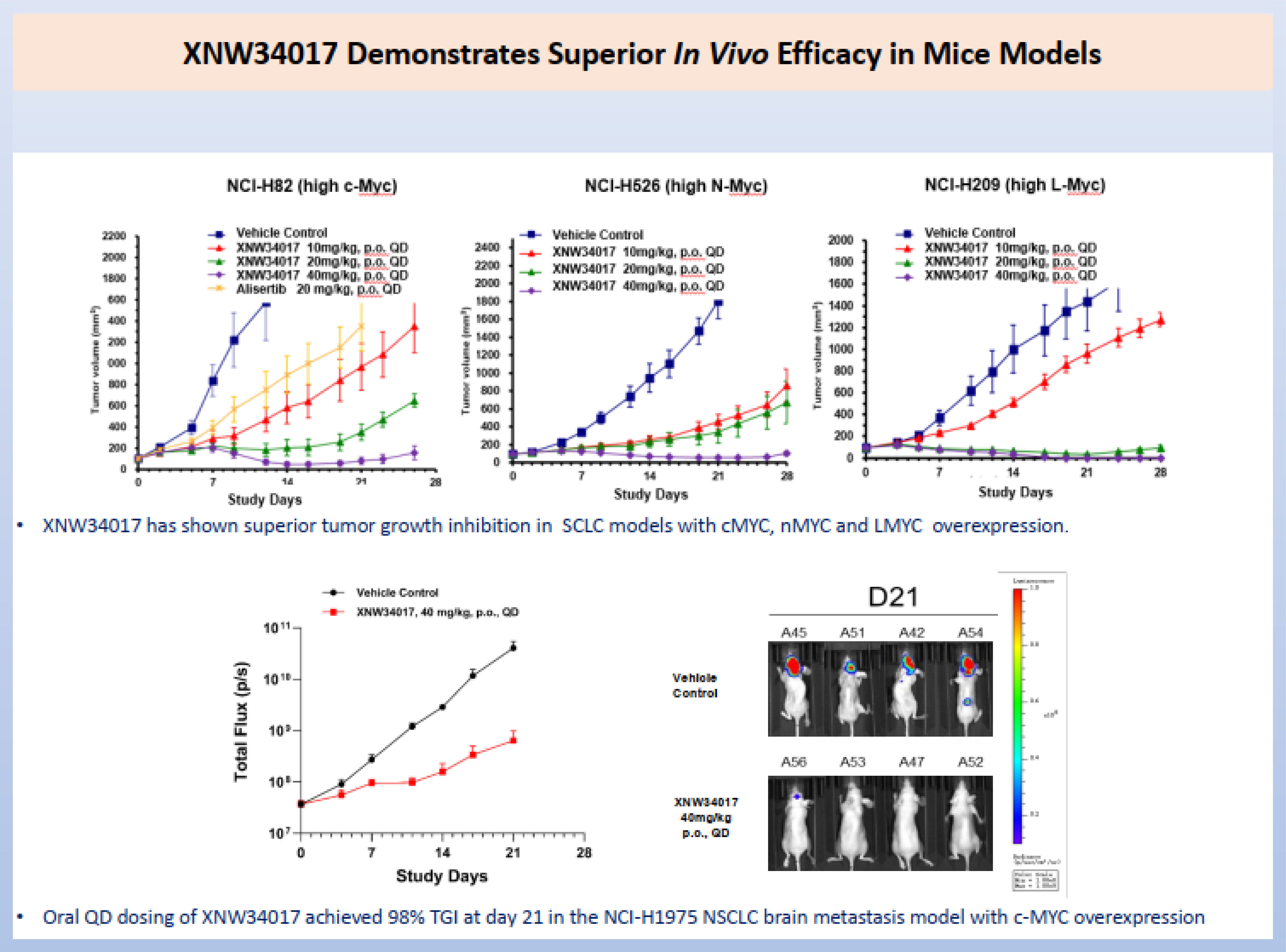
The systematically optimized investigation drug XNW34017 exhibited favorable pharmacokinetic profile, featuring an oral bioavailability exceeding 50% across mice, rats, and dogs, along with noticeable brain penetration. By selectively and efficiently degrading Aurora kinase A, this compound triggers downstream degradation of Myc family proteins. In multiple tumor models characterized by different Myc protein overexpression, XNW34017 demonstrated significantly superior tumor growth inhibition (TGI) compared to conventional Aurora kinase A inhibitors. Notably, in the NCI-H1975 non-small cell lung cancer (NSCLC) brain metastasis model with c-Myc overexpression, oral administration once a day of XNW34017 achieved a 98% TGI by Day 21, indicating its robust therapeutic potential against MYC-driven central nervous system (CNS) malignancies.
XNW27011 (CLDN 18.2 ADC)
Poster Title: Preclinical development of XNW27011, an antibody drug conjugate (ADC) targeting Claudin18.2 for treatment of CLDN 18.2-positive solid tumors,#1690
Highlights: XNW27011 is a novel antibody-drug conjugate (ADC) targeting Claudin 18.2 (CLDN18.2), featuring a potent cytotoxic payload conjugated to the antibody via a cleavable linker, with a drug-to-antibody ratio (DAR) of 8. Preclinical evaluations demonstrated that XNW27011 specifically binds to tumor cells with high CLDN18.2 expression. In various gastric and pancreatic cancer xenograft models—including both cell line-derived (CDX) and patient-derived (PDX) models—XNW27011 induced significant tumor regression at low dose levels.
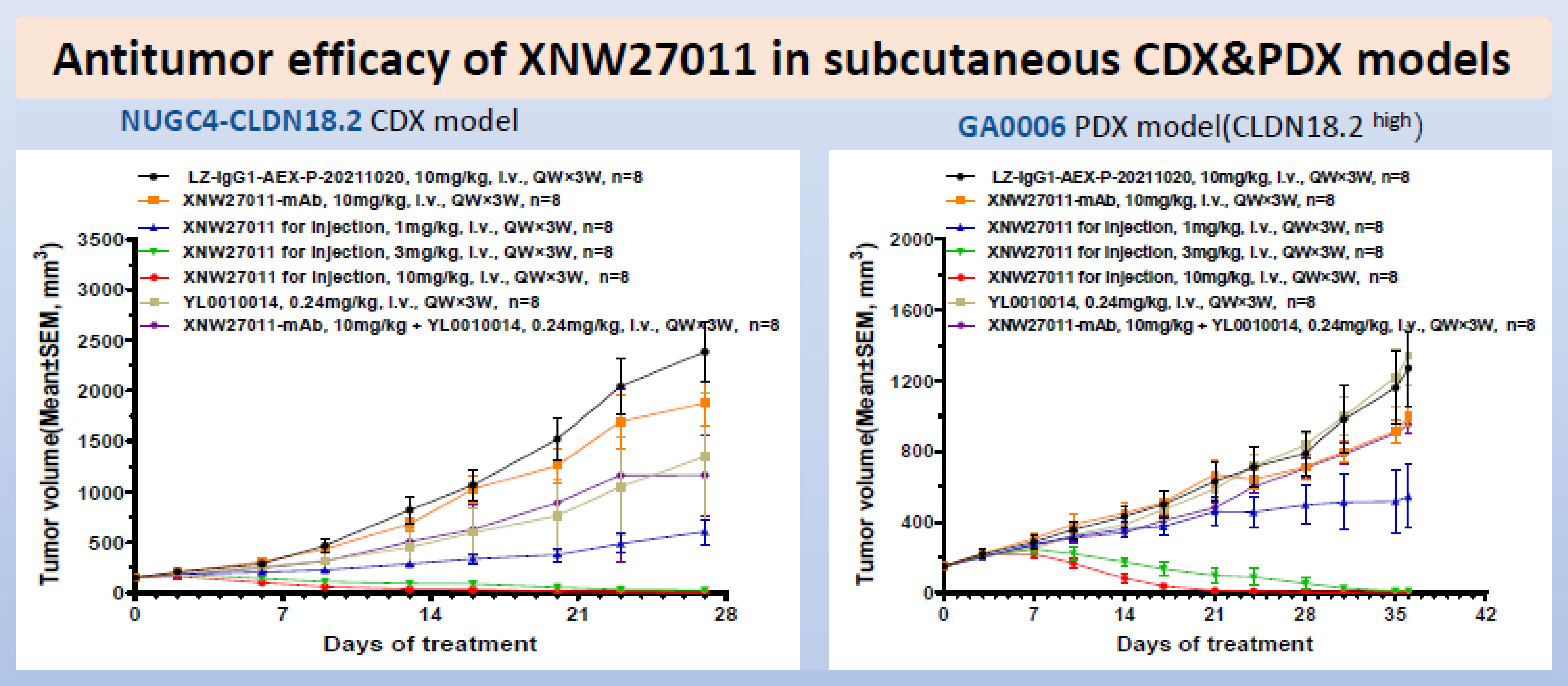
Pharmacokinetic and toxicological studies revealed that XNW27011 is highly stable in plasma and the systemic payload exposure of the ADC is low. The compound was well tolerated in both rodent and non-human primate models, with a wide therapeutic window; notably, no pulmonary or ocular toxicities were observed.
XNW28012 (TF ADC)
Poster Title: Preclinical development of XNW28012, an antibody drug conjugate (ADC) targeting tissue factor (TF) for treatment of solid tumors,#4437
Highlights: XNW28012 is a novel ADC targeting Tissue Factor (TF), comprising a highly potent DNA topoisomerase I inhibitor conjugated to the antibody via a highly stable, cleavable linker. TF is aberrantly overexpressed in a wide range of solid tumors and serves as an ideal ADC target due to its rapid internalization and efficient lysosomal trafficking upon antibody binding. Preclinical studies demonstrated that XNW28012 specifically binds to cell-surface TF, followed by linker cleavage within the tumor microenvironment and lysosomal compartments, which triggers the release of the cytotoxic payload to exert anti-tumor activity. In the tested multiple solid tumor models—including cervical, ovarian, and pancreatic cancers—XNW28012 exhibited significant, dose-dependent anti-tumor efficacy. Notably, the agent induced tumor regression at well-tolerated doses, outperforming the marketed TF-ADC therapy, Tivdak.
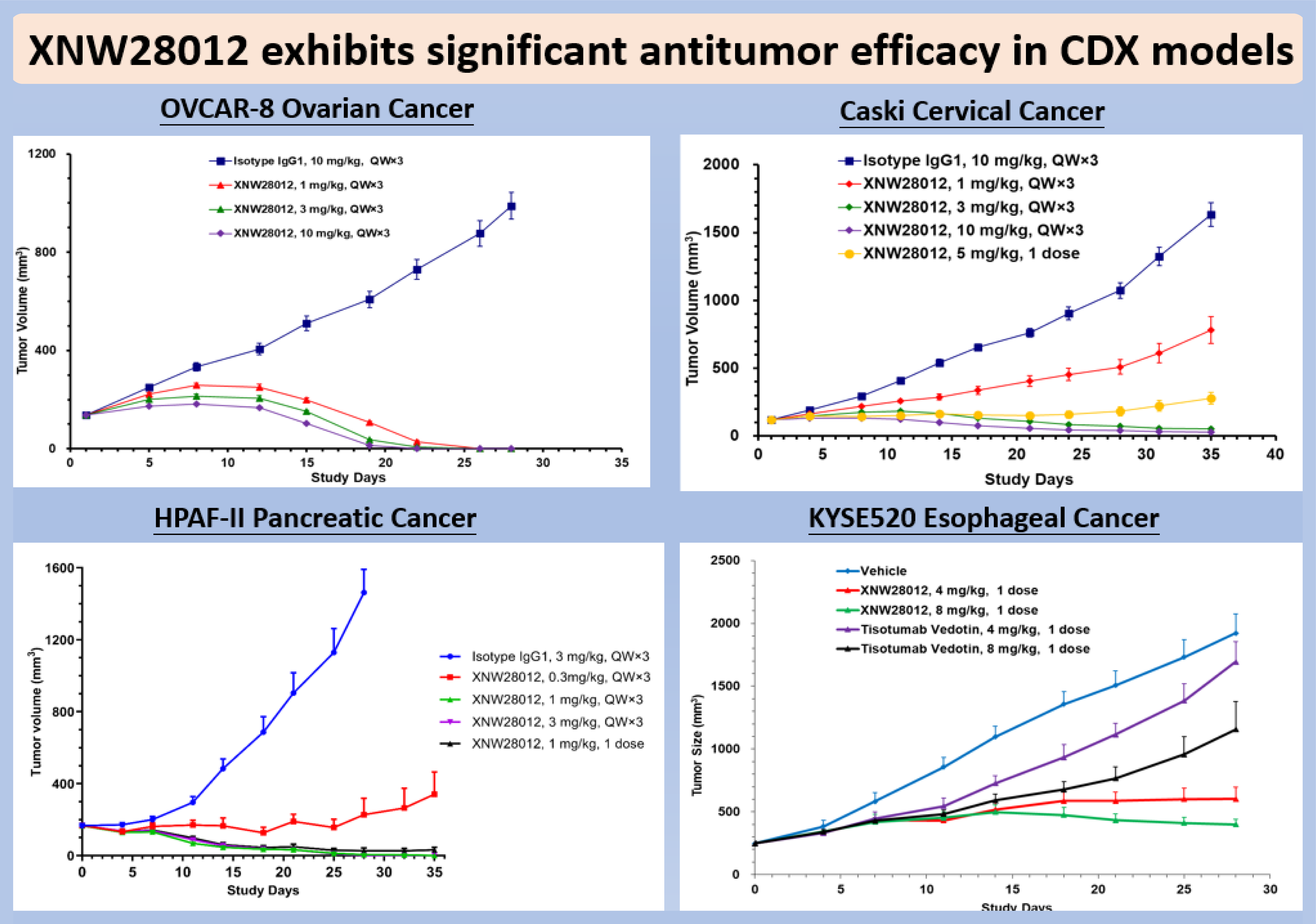
Pharmacokinetic studies demonstrated excellent plasma stability and low payload exposure of XNW28012. Toxicological studies revealed that XNW28012 was well tolerated in non-human primates with a wide therapeutic window; notably, no pulmonary or ocular toxicities were observed.





