
SUZHOU, CHINA--On February 27, 2026, Evopoint Biosciences announced its clinical results presentation of the EZH2 inhibitor Igermetostat (XNW5004), an independently developed Category 1 new chemical entity (NCE) by Evopoint, in combination with Enzalutamide in mCPRC at the 2026 American Society of Clinical Oncology Genitourinary Cancers Symposium (ASCO GU) for the first time, highlighting the emerging competitive advantage of the company in prostate cancer globally.
Abstract Title:A Phase 1b/2 study to evaluate the safety and efficacy of Igermetostat (XNW5004) in combination with Enzalutamide in patients with metastatic castration-resistant prostate cancer
Abstract No.:169
Presentation Type: Poster Presentation
Igermetostat in Combination with Enzalutamide Demonstrate Promising Efficacy and Safety in Patients with mCRPC who received Abiraterone previously.
In the Phase Ib/II clinical study evaluating Igermetostat in combination with enzalutamide in metastatic castration‑resistant prostate cancer (mCRPC), a total of 87 patients who were previously treated with novel hormonal therapies had been enrolled as of January 28, 2026. The combination demonstrated favorable safety and tolerability profile. No dose‑limiting toxicities (DLTs) was observed. 1200 mg BID for Igermetostat was selected as the recommended Phase II dose (RP2D).
56 patients who had received prior abiraterone therapy were included for efficacy analysis. The median follow‑up time was 17.5 months. The median radiographic progression‑free survival (rPFS) was 18.6 months in both the overall population who failed abiraterone and the patients who received Igermetostat at 1200 mg, BID. The median overall survival (mOS) in both patient groups was not reached yet (95% CI: 18.56–NR). At the 1200 mg dose level of Igermetostat, the 12‑month rPFS rate was 71.8%. Among the 16 patients with measurable disease at baseline, the objective response rate (ORR) was 25.0%, including 4 partial responses. Of the 35 PSA‑evaluable patients, 11 (31.4%) patients achieved a decline of PSA ≥50% from baseline.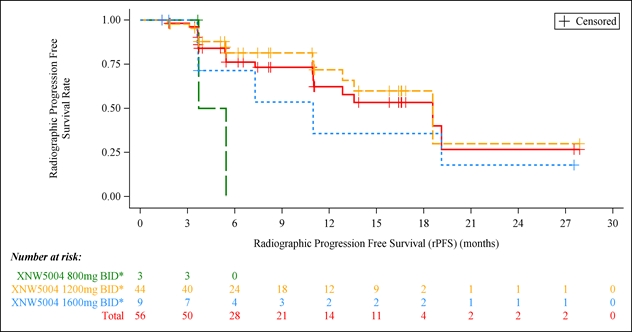
Figure 1. Radiographic Progression-Free Survival (rPFS) in patients previously treated with abiraterone
About Igermetostat (XNW5004)
Igermetostat (XNW5004) is a small‑molecule, highly selective EZH2 inhibitor with the best-in-class potential of efficacy worldwide. It is an EZH2 inhibitor that is at the forefront of global clinical development progress targeting EZH2. Igermetostat is classified as a Category 1 new chemical entity (NCE) in China. Current development strategy for Igermetostat is focused on indications of peripheral T‑cell lymphoma (PTCL), follicular lymphoma (FL), and metastatic castration‑resistant prostate cancer (mCRPC).
Igermetostat has demonstrated superior efficacy and favorable safety in lymphoma patients. It has been granted with Breakthrough Therapy Designation (BTD) from the CDE for indications of relapsed/refractory PTCL with two prior lines of systemic therapies and relapsed/refractory FL (EZH2 wild‑type) with at least three prior lines of systemic therapies. A pivotal study for Igermetostat in r/r PTCL as 3rd line therapy was initiated in November 2024. Meanwhile, two Phase III clinical studies of Igermetostat are ongoing, including the study in r/r PTCL as 2nd line therapy and in r/r FL (EZH2 wild‑type) after at least three prior systemic treatments.





